Lhasa Limited is pleased to expand its Complex Nitrosamine Dataset for the fourth consecutive year, a result of close collaboration with industry-leading organisations.
Offering new records to broaden the scope of complex nitrosamine understanding, the database is a direct result of active consortium meetings and the sharing of industry data.
The Lhasa data sharing initiative for complex nitrosamines reimagines experimental data, promoting shared records at the precompetitive stage to accelerate shared knowledge and industry progression. Data from 16 members in the pharmaceutical landscape has been collated and peer reviewed by Lhasa scientists to expand the Complex Nitrosamines 2026.1 dataset.
This year’s release marks another milestone made since the Valsartan crisis in July 2018, the detection of N-nitroso-varenicline and the inception of the Lhasa data sharing initiative in 2021.
Discover further details on the newly available Ames data records below.
Mutagenicity study donations for accurate assessments on novel substances
Identifying trends in compound complexity versus mutagenic potential has been made possible with Lhasa. All whilst maintaining the confidentiality of data records donated.
The latest update to the Lhasa data sharing initiative, Complex Nitrosamines 2026.1, has resulted in a unique database now containing:
- 563 new data records, bringing the total to 4554.
- 13 new substances, bringing the total number of substances to 148.
Data collated encompasses both in vitro bacterial mutagenicity data (data available on 143 nitrosamines) and in vivo genotoxicity data (data are available on 23 nitrosamines).
What does this mean in practice?
Here are four key ways in which the latest data records and the initiative have supported the assessment and management of nitrosamine impurities in pharmaceutical products:
- Confident risk assessments performed using an improved understanding of complex nitrosamines.
- Saved time and resources of our members by reducing the need for in-house testing.
- Informed regulatory decision-making using a strengthened, evidence-based, scientific judgement.
- Mutagenicity data contributed includes transgenic rodent and duplex sequencing assay results, for further regulatory-grade robustness.
To access the latest records in Complex Nitrosamines 2026.1, you must be a member of the Vitic solution and a participant in the consortium.
Uncovering new data, together
Regulatory agencies are evolving the N-Nitrosamine risk assessment requirements to ensure they highlight the potential presence of novel drug-like nitrosamines (NDSRIs) for which little or no publicly available toxicity data exists.
As we continue to expand the scope of the data-sharing initiative for complex nitrosamines, we want to ensure we meet industry needs.
To achieve safer and more efficient workflows in pharmaceutical development, as well as streamline regulatory submission consistency, the Lhasa data sharing collaborations, like Complex Nitrosamines 2026.1, will continue to grow.
From hosting annual in-person consortium meetings for shared research, to ensuring that data-led decisions are accessible across all novel compounds.
By joining, members can take advantage of data sharing:
- Reduced duplicate testing
- Quick identification of emerging trends
- Refined use of animal testing
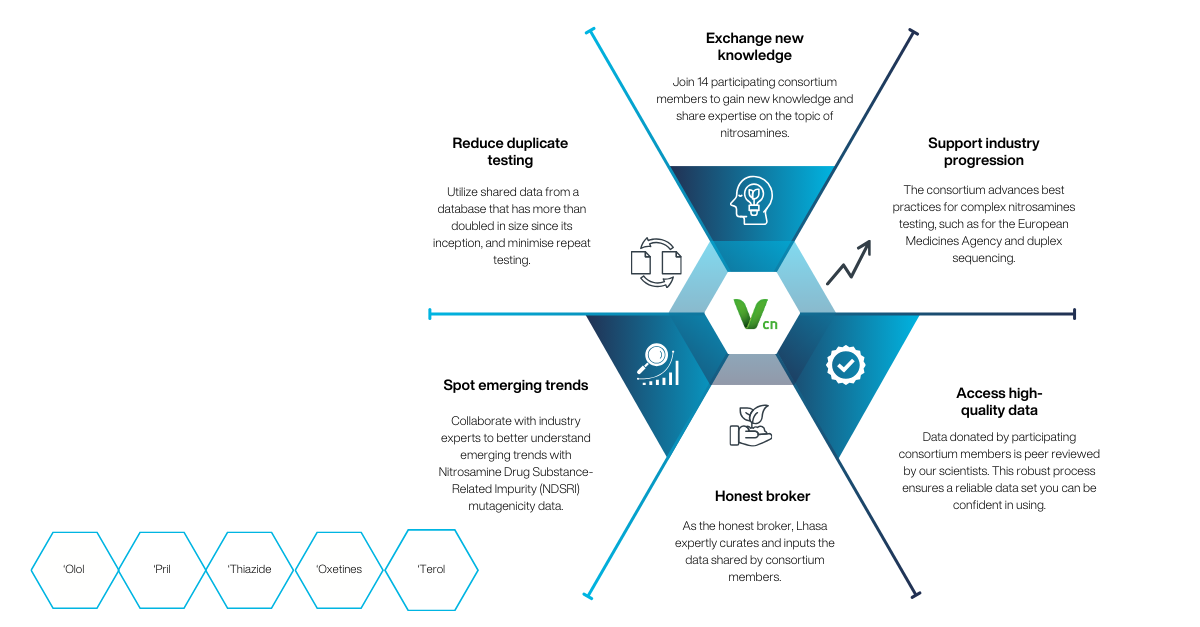
Drawing on comprehensive datasets from leading organisations, Lhasa delivers initiatives across various aspects of pharmaceutical risk assessments.
This effort in particular allows new data to facilitate the creation and validation of new structure-activity-relationship (SAR) models, better understanding of structural influences on mutagenicity, and more accurate toxicity predictions.
Joining the consortium
A larger set of real-world experimental results helps organisations learn from one another, collaboratively progressing efforts to positively impact human health.
Ready to unlock both Ames and in vivo genotoxicity data? Play a key role in tackling complex N-nitroso compounds and contribute your data by becoming a consortium member, all whilst maintaining the confidentiality of internal data.
Discover Complex Nitrosamines, the data sharing initiative from Lhasa Limited.
